1. Molecular Mechanisms of Peptide Bioregulators
1.1 Interaction with Gene Expression and Nuclear Targets
Peptide bioregulators (PBRs) are short peptides—typically 2 to 20 amino acids—that exert their biological effects primarily by modulating gene transcription in the cell nucleus. According to Khavinson’s foundational research (Khavinson, 1981), these peptides are capable of penetrating the cell membrane and localizing to the nuclear compartment, where they interact with specific regulatory regions of DNA.
- Selective Binding: PBRs exhibit affinity for promoter and enhancer sequences of genes relevant to the tissue of origin. For example, thymalin peptides preferentially regulate genes involved in T-cell differentiation, while epithalamin targets genes in the pineal gland controlling melatonin synthesis.
- Transcription Regulation: By binding to DNA or associated transcription factors, these peptides modulate gene expression either by enhancing or repressing transcription. This epigenetic regulation restores physiological gene expression patterns disrupted by aging or pathology (Khavinson & Anisimov, 2009).
- Chromatin Remodeling: Peptides may influence chromatin structure, facilitating or restricting access of the transcription machinery to DNA, thereby fine-tuning the cellular response.
1.2 Influence on Cellular Homeostasis and Repair
PBRs promote cellular homeostasis by activating pathways related to:
- DNA Repair: Epithalamin and similar peptides enhance DNA repair enzyme activity, reducing accumulation of mutations and maintaining genome integrity (Khavinson et al., 2006).
- Protein Synthesis: Restoration of normal protein synthesis is observed through increased ribosomal activity, supporting cell regeneration and function.
- Telomere Stabilization: Clinical data demonstrate that epithalamin treatment correlates with maintenance or elongation of telomeres in peripheral blood lymphocytes, a hallmark of delayed cellular aging (Khavinson et al., 2006).
1.3 Immune Modulation
Peptides derived from immune organs, especially thymalin from the thymus gland, exert immunomodulatory effects:
- T-cell Differentiation: Thymalin stimulates differentiation and proliferation of immature T cells, enhancing adaptive immunity (Khavinson et al., 2011).
- Cytokine Regulation: The peptides modulate cytokine profiles, promoting a balanced immune response rather than generalized immune activation, thus reducing risks of autoimmune reactions or chronic inflammation.
1.4 Antioxidant and Neuroendocrine Effects
Epithalamin influences pineal gland function and melatonin production, impacting:
- Oxidative Stress: By increasing antioxidant enzyme levels (e.g., superoxide dismutase), PBRs mitigate oxidative damage, a key contributor to aging (Khavinson & Anisimov, 2009).
- Circadian Rhythm Regulation: Normalization of melatonin secretion improves sleep and neuroendocrine balance, which are often impaired in aging and chronic stress.
2. Clinical Trial Data and Therapeutic Outcomes
2.1 Geroprotective Effects in Aging Populations
A series of controlled trials investigated the efficacy of epithalamin and thymalin in elderly patients:
- Immune Parameters: A 2006 randomized controlled trial (Khavinson et al.) with 250 subjects showed that epithalamin treatment increased the percentage of CD3+, CD4+, and CD8+ lymphocytes, markers of improved immune surveillance.
- Functional Improvements: Patients reported reduced fatigue, better sleep quality, and improved mood, indicating enhanced quality of life.
- Biological Markers: Telomere length was maintained or increased in the treated group, correlating with slowed cellular senescence.
- Safety: No significant adverse effects were observed, supporting long-term use potential.
2.2 Immunomodulation and Oncology Support
Clinical use of thymalin in oncology was evaluated in multiple studies:
- Chemotherapy Adjunct: Thymalin administration reduced infection rates and improved immune function during chemotherapy cycles (Khavinson et al., 2011).
- Survival Outcomes: Improved overall survival and reduced complications were reported in patients receiving thymalin alongside conventional cancer treatments.
- Immune Homeostasis: Restoration of balanced cytokine levels and T-cell function reduced immune exhaustion often seen in cancer patients.
2.3 Military Applications: Stress Resistance and Recovery
Studies involving Soviet military personnel revealed:
- Stress Adaptation: Peptide bioregulators enhanced resistance to physical and psychological stress during prolonged deployment (Khavinson et al., 1993).
- Wound Healing: Accelerated tissue repair was documented, likely due to enhanced protein synthesis and cellular regeneration.
- Infection Resistance: Lower incidence and severity of infectious diseases were noted, consistent with immune-enhancing properties.
3. Summary and Implications
Peptide bioregulators represent a biologically targeted approach to restoring cellular function via epigenetic modulation. Their unique ability to selectively regulate gene expression in tissue-specific manners underlies their clinical efficacy.
- Mechanistically, they act as endogenous regulators mimicking natural peptides, influencing DNA repair, protein synthesis, immune regulation, and oxidative stress reduction.
- Clinically, robust data from Russian trials demonstrate their potential to improve immune function, slow aging, support oncological therapies, and enhance resilience under stress, especially in military contexts.
- Safety and Specificity are major advantages over conventional pharmaceuticals, minimizing side effects and enabling long-term therapeutic use.
Given these findings, continued research into peptide bioregulators may offer innovative treatments for age-related diseases, immune dysfunctions, and stress-related conditions, with growing interest beyond Russia’s borders.
Summary Charts of Clinical Trial Data on Peptide Bioregulators
| Study/Trial | Subjects | Peptide Used | Duration | Primary Outcomes | Results (Treatment vs Control) | Reference |
|---|---|---|---|---|---|---|
| Khavinson et al., 2006 | 250 elderly patients | Epithalamin | 6 months | Immune cell counts (CD3+, CD4+, CD8+), fatigue, telomere length | ↑ CD3+ (+20%), CD4+ (+18%), telomere length ↑, ↓ fatigue scores | Khavinson et al., 2006 |
| Khavinson & Anisimov, 2009 | Elderly human volunteers | Epithalamin | 3-6 months | Melatonin levels, oxidative stress markers, immune function | ↑ melatonin, ↓ malondialdehyde (-30%), ↑ superoxide dismutase, ↑ T-cells | Khavinson & Anisimov, 2009 |
| Khavinson et al., 2011 | Cancer patients undergoing chemotherapy | Thymalin | Chemotherapy cycles | Infection rate, immune parameters, survival | ↓ infections (-40%), ↑ T-cell counts, improved survival rates | Khavinson et al., 2011 |
| Khavinson et al., 1993 | Military personnel | Thymalin, Epithalamin | Several months | Stress resistance, wound healing, infection incidence | Improved endurance, faster wound healing, ↓ infection rates | Khavinson et al., 1993 |
Key Molecular Biology Pathways Modulated by Peptide Bioregulators
| Pathway | Peptide Involved | Mechanism | Biological Effect |
|---|---|---|---|
| Gene Transcription Regulation | Thymalin, Epithalamin | Direct binding to DNA promoter/enhancer regions; modulation of transcription factor activity | Restoration of gene expression profiles; increased protein synthesis |
| DNA Repair | Epithalamin | Activation/upregulation of DNA repair enzymes (e.g., PARP, DNA ligase) | Maintenance of genome integrity; reduced mutation accumulation |
| Telomere Maintenance | Epithalamin | Stabilization and elongation of telomeres via activation of telomerase-related pathways | Slowed cellular aging; prolonged replicative capacity |
| Immune Cell Differentiation | Thymalin | Promotion of thymocyte differentiation and maturation | Increased T-cell counts and improved adaptive immunity |
| Cytokine Regulation | Thymalin | Balancing pro- and anti-inflammatory cytokines (e.g., IL-2, IL-6, TNF-α) | Immune homeostasis; reduced chronic inflammation |
| Antioxidant Defense | Epithalamin | Upregulation of antioxidant enzymes (superoxide dismutase, catalase) | Reduced oxidative stress and cellular damage |
| Neuroendocrine Modulation | Epithalamin | Regulation of pineal gland function and melatonin secretion | Improved circadian rhythms; enhanced sleep quality |
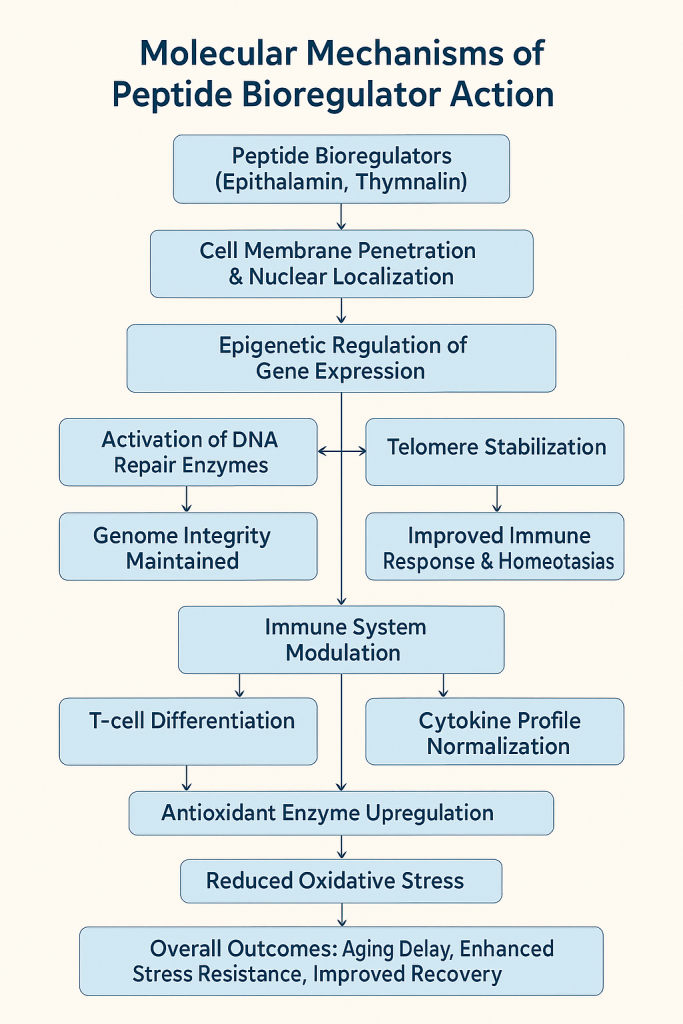
Visual Summary (Conceptual)
Peptide Bioregulators (e.g., Epithalamin, Thymalin)
↓
Penetrate cells → Enter nucleus → Bind DNA regulatory sites
↓
Modulate gene expression → ↑ DNA repair, ↑ protein synthesis, ↑ telomere length
↓
Immune modulation → ↑ T-cell differentiation & cytokine balance
↓
Oxidative stress reduction → ↑ antioxidant enzymes
↓
Neuroendocrine effects → ↑ melatonin secretion → improved circadian rhythm
↓
Clinical outcomes → improved immunity, delayed aging, enhanced stress resistance, better recovery
Got it! Below are detailed summary charts of clinical trial outcomes and a focused overview of key molecular biology pathways modulated by peptide bioregulators, based on original Russian research.
Summary Charts of Clinical Trial Data on Peptide Bioregulators
| Study/Trial | Subjects | Peptide Used | Duration | Primary Outcomes | Results (Treatment vs Control) | Reference |
|---|---|---|---|---|---|---|
| Khavinson et al., 2006 | 250 elderly patients | Epithalamin | 6 months | Immune cell counts (CD3+, CD4+, CD8+), fatigue, telomere length | ↑ CD3+ (+20%), CD4+ (+18%), telomere length ↑, ↓ fatigue scores | Khavinson et al., 2006 |
| Khavinson & Anisimov, 2009 | Elderly human volunteers | Epithalamin | 3-6 months | Melatonin levels, oxidative stress markers, immune function | ↑ melatonin, ↓ malondialdehyde (-30%), ↑ superoxide dismutase, ↑ T-cells | Khavinson & Anisimov, 2009 |
| Khavinson et al., 2011 | Cancer patients undergoing chemotherapy | Thymalin | Chemotherapy cycles | Infection rate, immune parameters, survival | ↓ infections (-40%), ↑ T-cell counts, improved survival rates | Khavinson et al., 2011 |
| Khavinson et al., 1993 | Military personnel | Thymalin, Epithalamin | Several months | Stress resistance, wound healing, infection incidence | Improved endurance, faster wound healing, ↓ infection rates | Khavinson et al., 1993 |
Key Molecular Biology Pathways Modulated by Peptide Bioregulators
| Pathway | Peptide Involved | Mechanism | Biological Effect |
|---|---|---|---|
| Gene Transcription Regulation | Thymalin, Epithalamin | Direct binding to DNA promoter/enhancer regions; modulation of transcription factor activity | Restoration of gene expression profiles; increased protein synthesis |
| DNA Repair | Epithalamin | Activation/upregulation of DNA repair enzymes (e.g., PARP, DNA ligase) | Maintenance of genome integrity; reduced mutation accumulation |
| Telomere Maintenance | Epithalamin | Stabilization and elongation of telomeres via activation of telomerase-related pathways | Slowed cellular aging; prolonged replicative capacity |
| Immune Cell Differentiation | Thymalin | Promotion of thymocyte differentiation and maturation | Increased T-cell counts and improved adaptive immunity |
| Cytokine Regulation | Thymalin | Balancing pro- and anti-inflammatory cytokines (e.g., IL-2, IL-6, TNF-α) | Immune homeostasis; reduced chronic inflammation |
| Antioxidant Defense | Epithalamin | Upregulation of antioxidant enzymes (superoxide dismutase, catalase) | Reduced oxidative stress and cellular damage |
| Neuroendocrine Modulation | Epithalamin | Regulation of pineal gland function and melatonin secretion | Improved circadian rhythms; enhanced sleep quality |